ABSTRACT
Background/Aim
Toxicity information of any raw milk is both rare and scarce, and it’s traditional consumption as raw; especially from Arabian camels haven’t been sufficiently validated. This research report was aimed to assess both raw camel milk’s safety through oral toxicity determination via both monitoring of renal function indices and observance of rat mortality, deformity, abnormal behavior or signs of toxicity manifestation when the camel milk was gavagely dispensed to rats via oral route.
Methodology
Fresh Arabian camels’ raw milk and other standard tools normally used for toxicity assessment using OECD 425 Up and Down procedural guidelines were employed for both acute (immediate and up to 2 days duration) and sub chronic gavage oral administration (that lasted for 4 weeks) to L-NAME induced hypertensive rats at (50 mg/kg/day) and compared with their normal, hypertension positive (amlodipine at (10 mg/kg/day) and negative (received only L-NAME at 50 mg/kg/day) controls. The raw camel milk tests were rats fed orally with it by gavage and dosed as CM1, CM2 and CM3. CM1, 2 and 3 was 100, 300 and 500 mg/kg/day of milk samples concurrently administered for 28 days with chronic hypertension.
Results/Conclusion
The absence of any rat mortality and non-exhibition of signs of toxicity by any rat involved in the study as well as null and void abnormality in the renal evaluation results attest to the zero-toxicity status of raw camel milk. Urea and creatinine renal results for the tests were lower than their positive and very higher in negative control peers. An oral LD50 was greater than 5000 mg/kg. Conclusively, it can be said that raw camel milk is both safe and toxic-free based on the obtained results.
INTRODUCTION
The flurry of several and extensive research on Camel Milk (CM); and acknowledgment of same for its contribution to human diet and crucial roles it plays in nutrition have earned it many accolades, the prominent one being described as ‘white gold of the desert’ (Shakeelet al., 2022), obviously considering that camels’ usual habitat is desert and CM possessed rich nutrients and diverse content. This rich nutrient value is due to functional ingredients (Alebieet al., 2017) contained therein.
The superiority of CM is unique and distinct when compared with other milk sourced from cow and other tamed animals dwelling with humans; in terms of nutritional composition (Hoet al., 2022), which made it to thus have superb qualities against human milk in terms of content variety and high amounts of bioactive compounds.
Contextually thus, evaluating nutritional composition of CM it is both safe and ideal to quote that it is above average and adequate with comparable advantage in contrast to some domestic animals’ milk (Dogondaji et al., 2023a).
Interestingly from folklorist accounts the therapeutic properties and possible deterrence of diseases offered by CM were recognized by the earlier generations allowing them including it as part of their daily life usages (Mihicet al., 2016). The following diverse ailments that include allergy, diabetes mellitus, renal disorder and virus or toxin-elicited liver inflammation were all been treated with promising effect in some recent previous studies with camel milk (Shakeelet al., 2022, Kamal-Eldinet al., 2022).
In particular some hepatoprotective effect of raw CM was demonstrated against chemical induced hypertension in some studied rats (Dogondajiet al., 2024); same too was has hypotensive roles in another earlier controlled study (Dogondaji et al., 2023b). More recently connected to the pursuit of immune-boosting foods by consumers during period of COVID-19 pandemic, these important therapeutic properties of camel milk were further underlined (Nagyet al., 2021).
Apparent health benefits of camel milk and its trending health consciousness by its consumers has made CM to gained widespread market exposure and commercialization which has align with continuous increase in the rate of non-communicable diseases morbidity (Nagyet al., 2021). Presently there are many processed products from camel milk available in many countries in different forms including its coffee, ice cream, cheese, pulverized or fine-grained milk all made possible through modern industrial processing (IMARC, 2021, Desoukyet al., 2019).
An apparent increased risk of aflatoxin contamination is however very much possible with other food sources and raw milk consumption irrespective of its origin, this possibility pointing to utmost need for evaluation of raw CM in addition to other food sources. Aflatoxins are natural toxins that may induce adverse effects on many living organisms including crops, other animals and humans which may results in some economic losses and many ailments (Hussain and Anwar, 2008, Akinyemiet al., 2022).
An estimate from the Food and Agricultural Organization; FAO, have put it at one-quarter of all food staple produced in the world are being polluted with mycotoxins. The natural milk produced from animals constitutes part of this global food. In fact, an AFM1 aflatoxin is found present in any milk. Fungus species such as Aspergillus flavus and Aspergillus parasiticus, were those responsible for proliferation of Aflatoxins.
The consumption of raw camel milk although, was linked to Brucella melitensis infection (Dubeyet al., 2016), but this possibility of raw camel milk consumption causing any disease was later dismissed from another account (Akinyemiet al., 2022).
Why many consumers of natural sources insist on having it as raw was to evade the possible loss or destruction of biological and immunological properties inherent in many milk that tend to be affected by the milk preservation processes; boiling or other processing stages might lead to such loss (CFSPH, 2018).
The study reported here aimed to assess potential toxicity of raw camel milk oral gavage administration acutely and in sub-chronic mode irrespective of raw milk microbial contamination status. Also, some renal indices obtained due to milk treatment were compared with their control equivalents in normal, positive and negative groupings. These data provide additional insights in respect of raw camel milk safety.
METHODOLOGY
Chemicals and Reagents
All the chemicals used during this study were all analytical grade and of purest quality available.
The Nitric Oxide Synthase; NOS inhibitor; [L-NAME] -Nitro-L-Arginine Methyl Ester that was used for hypertension induction was purchased from German Chemical Company, (Sigma Aldrich). The kits that were used for the determinations of conjugated bilirubin and alkaline phosphatase were purchased from Abnova (Taiwan). The Manufacturer’s instructions for the usage of the kits were strictly followed; including their storage at 2-8ºC and were left for at least 1 hr at room temperature prior to their initial use. De-ionized water and other chemicals used during this study were also of analytical grade.
Raw Camel Milk (CM) sample collection
Five (5) healthy lactating female camels (Camelus dromedarius) from Kwakwalawa village; Sokoto State, Nigeria whose ages were 5-10 years old was sources for the needed milk samples that was collected by hand milking following proper sterilization of the Camels udders. 5 L equivalent of milk from each female camel were collected by experienced and skilled camel attendant during morning time. Samples were first collected into properly labeled plastic containers [A-E] and were then carefully transferred into another labeled suitable sterile glass bottles (vials). Immediately, the obtained samples were transported via cold chain medium to the Research laboratory of the Usmanu Danfodiyo University, Sokoto and refrigerated at -20ºC until required for further use.
Ethics Statement
Animal studies were performed in accordance with recommended advice for the Use and Care of Animals in the Laboratory by the (OECD, 2008) procedures. Permission was granted by the University Research Ethics Committee of the Centre for Advance Medical Research and Training; CAMRET of the University (UDUS) via approval letter UDUS/UREC/2019/020 for this study to use albino rats.
Experimental Animals
Overall, forty-four albino rats (Wistar strains) of both sexes were used in both acute and sub-chronic toxicity research. Required albino rats used in this study were procured on purchase from the Animal House of Department of Pharmacology and Toxicology of Faculty of Pharmaceutical Sciences, College of Health Sciences, Usmanu Danfodiyo University, Sokoto, Nigeria. Weights of all the rats ranged between 150-250 g and they were acclimatized in wire mesh cages for 2 weeks to adapt to the new laboratory environment where they were caged before the commencement of conducted experiments. Rats were randomly divided and assigned into properly labeled cages and kept separately according to their sex to protect females from conceiving during study period. Animal feed and drinking water were provided ad libitum.
On premise of Up and Down Procedure; the acute toxicity test was performed in accordance with Organization for Economic Co-operation and Development; OECD 407 document Guidelines 407 with 6 rats chosen randomly for each sub group. Tests were conducted via 2 steps for; (i) 2000 mg/kg and (ii) 5000 mg/kg phases and each rat involved in the 2 steps represent its group with each among them receiving appropriate single oral dose of either 2000 or 5000 mg/kg of CM and their controls similarly given same volume content of distilled water (1 rat for each category) that made 14 rats in all for acute study. In sub chronic toxicity studies; there were (3) major sub groupings as A, B and C in which each sub group also has 5 rats housed in their labeled cages. Group D was the control set and it had 1 rat for each of A, B and C, making total of 18 rats. Detailed procedure was in the subsequent narration beneath
General procedure
Rats were handled in both acute and sub-chronic stage in a simple similar way. The feeding cannula used to give oral gavage of samples during animal handling was held by right hand and rat was held with left hand. The neck and head of rat were kept in straight posture to allow cannula enter rat mouth easily. After insertion of the cannula into any rat’s mouth by about 5 cm deep, it was then pushed slowly to allow cannula content being delivered into the rat body. Rats that agreed gently with minimal or no resistance, were given the whole milk content slowly. Those rats that resisted and engaged in struggle, their cannula was withdrawn paused a little and gently reworked.
Clinical monitoring started immediately after the simultaneous single oral dose CM gavage treatment then followed after 4 hr. From then after every 8 hr, the rat will be monitored to last for at least 7 days.
Reconstitution of CM oral doses for administration
Bulk refrigerated CM samples (A-E) were shaken to become uniform and homogenized; then poured separately in 5 big volumetric flasks each flask bearing appropriate sample identity as A-E, from where further measurements for subsequent analyses were made. Pipette and graduation flasks were used to measure and reconstitute CM samples to the required concentration dosages of 2000 mg/kg and 5000 mg/kg used in acute oral study. From same bulk homogeneous sample, appropriate volumes were taken and reconstituted into 100, 300 and 500 mg/kg concentration doses in the subsequent sub-acute toxicity studies where it was orally administered by gavage to assigned rats based on their determined body weights during continuous daily oral administration for up to 4 weeks.
Acute oral toxicity evaluation
In acute oral toxicity evaluation of raw camel milk; standard methods that complied with 2008 Organization for Economic Co-operation and Development; OECD 407 document 425 guidelines were followed. Thus, CM single oral dose at 2000 mg/kg were administered to 6 group of rats (each rat representing its group) at grace observation period of 48 hr and the rats were monitored for changes in their behavior; distress and toxic symptoms manifestation, abnormal pattern of food and water consumption and rat mortality from treatment-related incidence. Observation began immediately within first 4 (critical) hr, then at 8 and 16 hr for up to 24 hr period and there was their continuous close monitoring until next day (48 hr). Control rats used per each group was given similar volume of distilled water and was also monitored accordingly.
Similarly, in another separate study same CM single oral dose at 5000 mg/kg body weight were administered to another set of 6 groups of rats for similar grace observation period of 48 hr and those rats too were also observed for obvious signs of toxicity and symptoms of distress, food and water consumption pattern and rat mortality. The control rats for these 2nd set for each group were also treated similarly with similar volume of distilled water as done with the test. Observations made were recorded and documented appropriately.
Sub chronic toxicity evaluation
There were (3) sub groups of rats housed in respective cages A, B and C; with each cage having 5 rats which were all treated according to 407 guidelines. These groups of rats were all having both hypertension and treatment. Rats in treatment group received appropriate CM single dose once daily by gavage. In this sub chronic toxicity phase; the repeated CM single oral doses were daily administered at (100, 300 and 500 mg/kg/day) simultaneously to the different concentration dosing groups together with chronic hypertension induction with dissolved L-NAME solution at (50 mg/kg body weight) uniformly during morning time between 10-11 am for 28-days. Group A received 100 mg/kg body weights/day of raw CM, Group B received 300 mg/kg body weights/day and Group C received 500 mg/kg body weights/day respectively immediately after given dissolved L-NAME treatment.
Control groups
Another rat groups simultaneously treated were (i) normotensives- devoid of any treatment or intervention except being given distilled water (ii) hypertensive negative and (iii) positive controls respectively.
Positive control received amlodipine (10 mg/kg body weights/day) in addition to their hypertensive induction (50 mg/kg/day of dissolved L-NAME) and negative controls were only inducted with hypertension via dissolved L-NAME solution only at (50 mg/kg body weight/day) lacking any intervention or treatment.
Rats in sub chronic toxicity studies were closely monitored two times during the day for immediate abnormal clinical signs after treatment followed by the critical 4 hr post treatment for manifestation of signs and symptoms of distress and toxic effect, their movement pattern in cages, and their food/water consumption and rat mortality rate from beginning to end of study were all monitored and recorded appropriately.
The whole rats groups in the entire study were all monitored for change in body weight and weights obtained was recorded every day prior to treatment, also once a week within period of study and on final day of research when the rats were sacrificed. On 29th day of study, after overnight fast (though rats were not denied free access to water for their drinking) all rats were slaughtered and their blood samples collected through retro-orbital bleeding into specialized test tubes for further biochemical analysis.
LD50 Determination
LD50 is the median lethal dose capable of killing half the number of animals receiving treatment in up and down procedure of toxicological studies and it is calculated to be above the upper limit of the tested dose used in intoxicating animals by oral administration.
Renal parameters assay
Serum preparation
After ten hr of fasting and one complete day post the final treatment, on the 29th day rats were killed through humane approach by light anesthesia administration with anaesthetic ether (ether/chloroform mixture dipped with cotton wool) which results in eliciting rapid absence of mind and subsequent death without much distress and or pain. Rats were then slaughtered. Their blood samples were taken from retro orbital plexus of each rat. Tubes containing anticoagulant were used for the rats’ blood samples collection. The samples collected were centrifuged immediately at 4000 rpm for 10 min duration thereby separating the needed serum for biochemical indices measurement and plasma obtained was kept at -40ºC until it is required for further analysis.
Kidney Integrity Function Analyses
Assessment of Urea was based on combined methods of (Wybengaet al., 1971; Wills and Savory, 1981), creatinine was estimated through method of Jaffe’s reaction developed by (Krollet al., 1987). The (Uriyo and Singh, 1974) method was used for the determination of renal electrolytes (sodium, potassium, chloride and bicarbonate).
Experimental Design in Brief
Blatant raw camel milk consumption has surged upwards in recent times; albeit for different reasons by diverse population have informed this decision; first to assess toxicity status of raw camel milk. If safe, then other ascribed attributes for its consumption can be carefully evaluated. This research interest primarily was focused on the determined LD50. Current determined LD50 dose in acute study was greater than 5000 mg/kg which have informed further decision in calculating subsequent doses to be used in sub-chronic oral toxicity tests. Three different doses in sub-acute study were arrived at (500, 300 and 100 mg/kg body weights) representing higher, medium, and lower doses, respectively, which were explored in the subsequent sub-acute study that followed.
Statistical analysis
The Levene’s test for homogeneity of variance and one-way Analysis of Variance (ANOVA) using the SPSS software was deployed for the analysis of the statistical generated data from renal investigations. The graphs were drawn by GraphPad Prism 5.0 (GraphPad Prism, San Diego, USA). Multiple comparisons of the obtained data expressed as (mean±SEM) was done by Tukey’s tests. When p<0.05 and or when chi-square approximation value is less than 0.0001 (<0.0001) the observed differences were taken to be statistically significant.
RESULTS AND DISCUSSION
Acute oral toxicity evaluation
The result of acute oral toxicity testing determined by observational monitoring of all the rats for displaying of any sign or symptom of toxicity was presented in Table 1. No toxic symptoms were observed in the entire rats used in this phase. The 3 broad sub groups in this study were (a) rats that received 2000 mg/kg raw CM and (b) those that were administered 5000 mg/kg as well as (c) their control group respectively. These results are as shown below.
| Single oral dose (mg/kg b.wt.) | Category | Signs of toxicity | Mortality |
|---|---|---|---|
| 2000 | E1 | Nil (-) | Nil (-) |
| 5000 | E2 | Nil (-) | Nil (-) |
| 5000 | NC | Nil (-) | Nil (-) |
Sub chronic toxicity evaluation
The obtained results for sub-chronic oral administration of raw camel milk to the experimental rats have also not indicated occurrence of any death among them that could be tied to treatment-related incidence during the 28-day study period. The absence of clear changes in general signs of clinical monitoring which include the rats’ response when it is being handled, their gait and or their posture all of such equally showed that the administered dose under study; is relatively not toxic, albeit there was diarrhea and slight depression within CM3; which is the group that received the highest dose of the sample under study.
Result of sub-chronic toxicity evaluation for 28 days at different graded concentrations is summarized in Table 2.
| Category Mortality | Number | Behavioural | SOT | Change in Feeding pattern |
|---|---|---|---|---|
| A | 5 | No (-) | No (-) | No (-) |
| B | 5 | No (-) | No (-) | No (-) |
| C | 5 | 1 (+) | No (-) | No (-) |
| NC | 5 | No (-) | No (-) | No (-) |
| HNC | 5 | No (-) | No (-) | No (-) |
| PC | 5 | No (-) | No (-) | No (-) |
Raw CM LD50 Determination
The determined oral LD50 of raw CM was above 5000 mg/kg body weight. It was arrived at after the experimental rats had survived beyond not only 2 days but also 14 days of observation from any post-treatment related incidence.
Food Consumption, Drinking water and Body Weight Assessments
There was no observed deviation from the normal pattern of food consumption or drinking of water by the entire rats in this research. Data that recorded amount of feed given before and during the research was recorded but it was not included here since there is no apparent significant statistical change that was observed. This has further illustrated that raw camel milk does not have any obvious effect on the rats’ food consumption.
The Renal marker enzymes
Renal Function Integrity Tests of rats given raw camel milk.
The Renal Function Tests results of rats given raw camel milk were graphically represented in Figures 1–6 as shown for each as captured below.
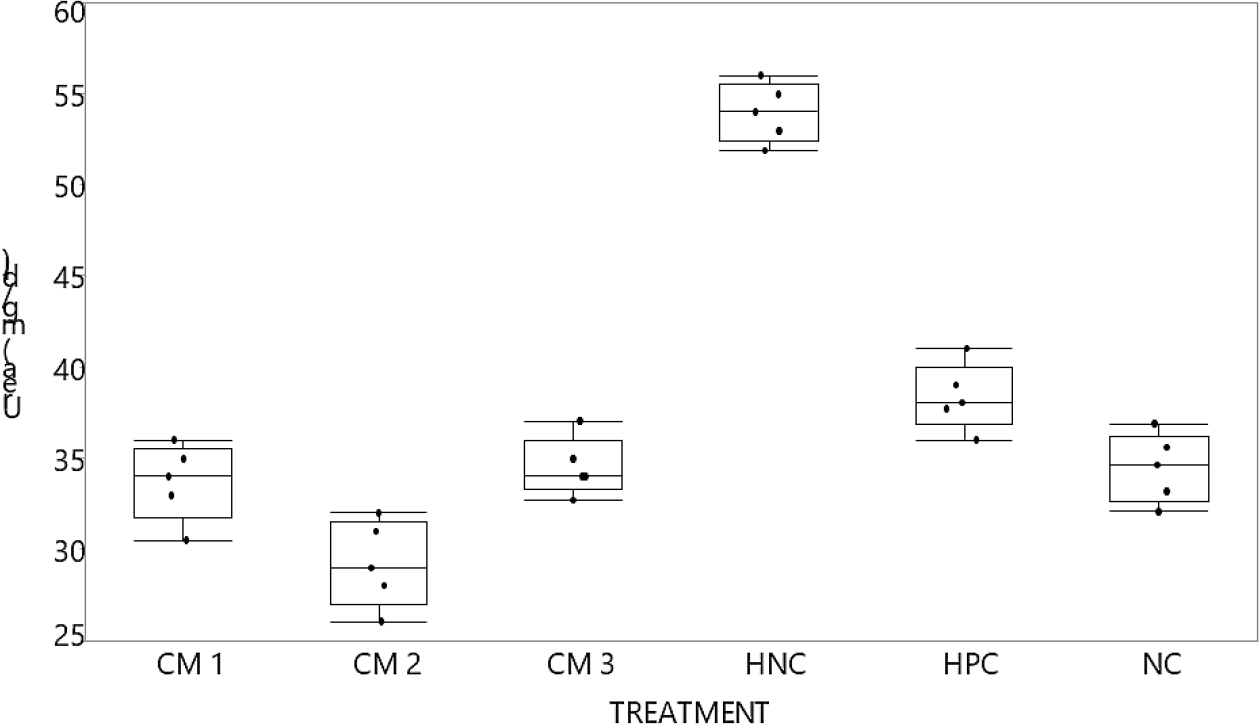
Figure 1:
Post hoc graph of Urea (g/dL). Urea (mg/dL) values obtained for CM concentrations compared to controls. Control group:-HPC, there is p-value statistical significance for CM3 only at 0.0005* and for 1-way test chi square approximation at 0.0002*. NC:-normal control; PC:-positive control (ref. group) amlodipine 10 mg/kg/day; HNC:- Negative control for hypertension (groups given L-NAME only [50 mg/kg/day]); L-NAME:-nitro-l-arginine methyl ester; CM1:-given raw camel milk at (100 mg/kg/day); CM3:-given raw camel milk at (300 mg/kg/day); CM5:-given raw camel milk at (500 mg/kg/day).
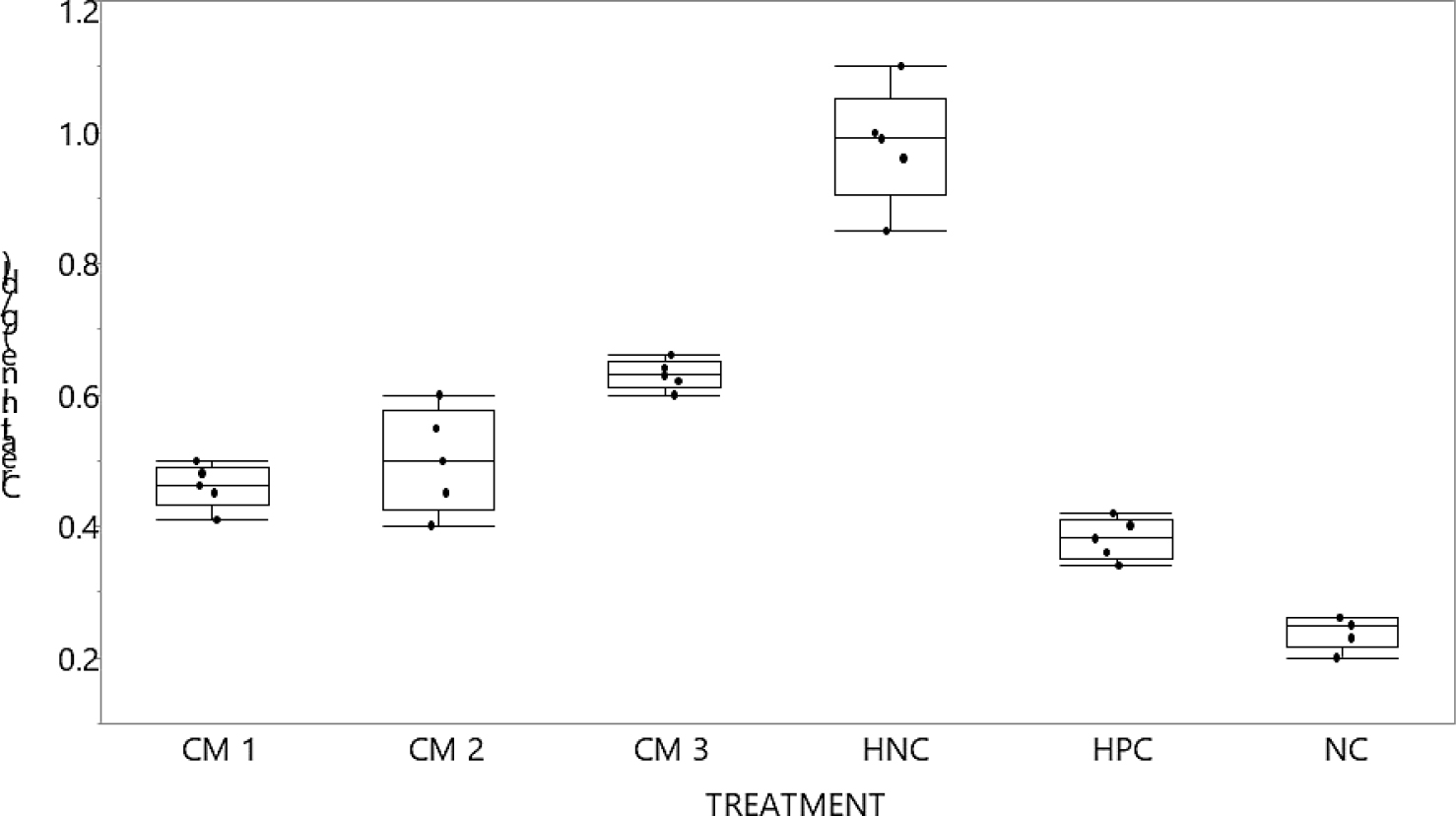
Figure 2:
Creatinine (g/dL) graph compared. Various controls compared with different CM concentration doses. Non-parametric comparison with positive control using Dunn Method for joint ranking. Control group:-HPC, there is no statistical significance for p-value but there is for 1-way test chi square approximation at <0.0001*. NC:-normal control; PC:-positive control (ref. group) amlodipine 10 mg/kg/day; HNC:- Negative control for hypertension (groups given L-NAME only [50 mg/kg/day]); L-NAME:-nitro-l-arginine methyl ester; CM1:-given raw camel milk at (100 mg/kg/day); CM3:-given raw camel milk at (300 mg/kg/day); CM5:-given raw camel milk at (500 mg/kg/day).
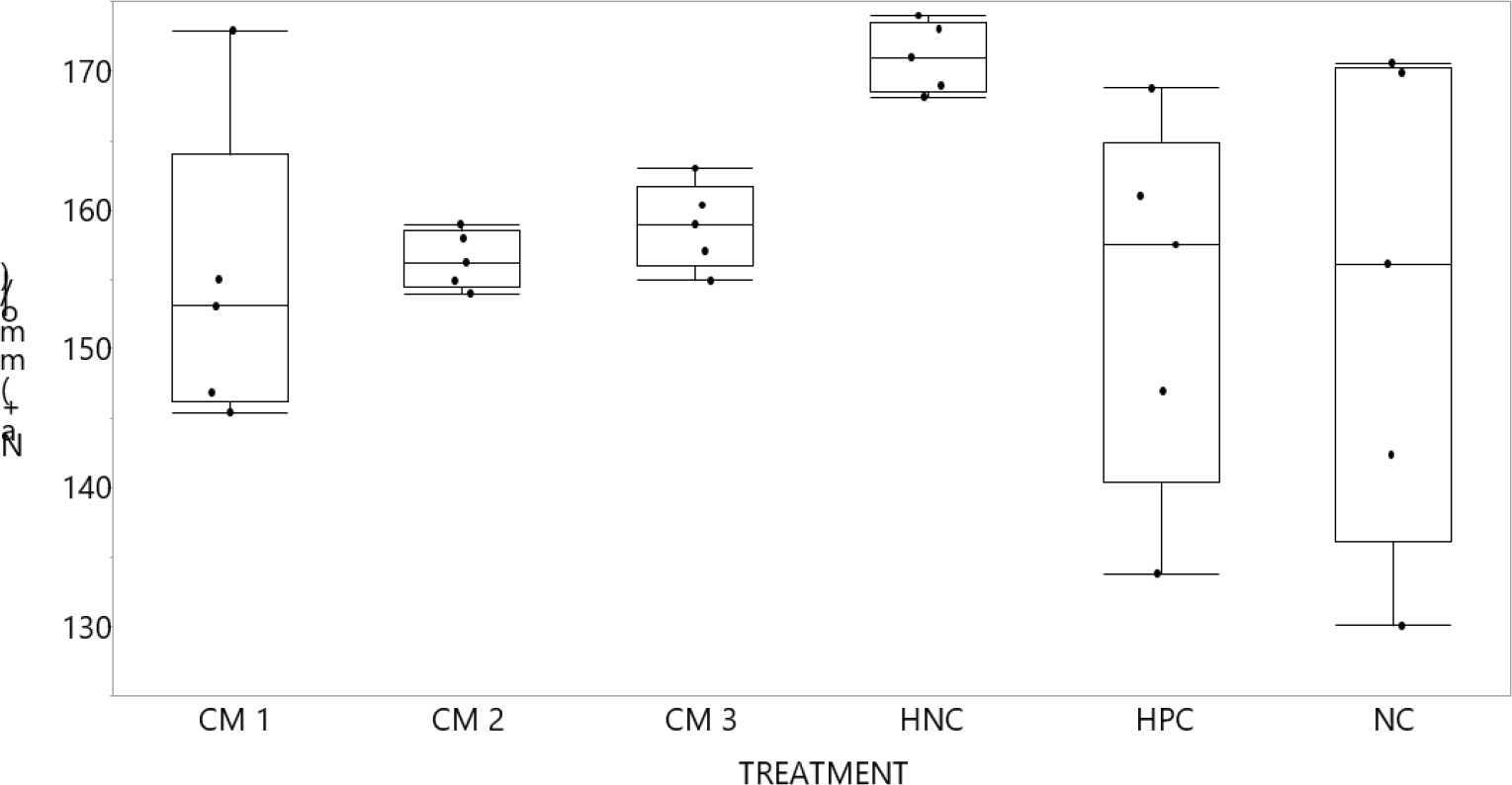
Figure 3:
Sodium ions; (Na+) (mmol/l) graph compared post interventions. Milk treatments compared with controls. Non-parametric comparisons with positive control using Dunn Method for joint ranking. Control Group=HPC. Na+:-Serum sodium ions; NC:-normal control; PC:-positive control (ref. group) amlodipine 10 mg/kg/day; HNC:- Negative control for hypertension (groups given L-NAME only [50 mg/kg/day]); L-NAME:-nitro-l-arginine methyl ester; CM1:-given raw camel milk at (100 mg/kg/day); CM3:-given raw camel milk at (300 mg/kg/day); CM5:-given raw camel milk at (500 mg/kg/day).
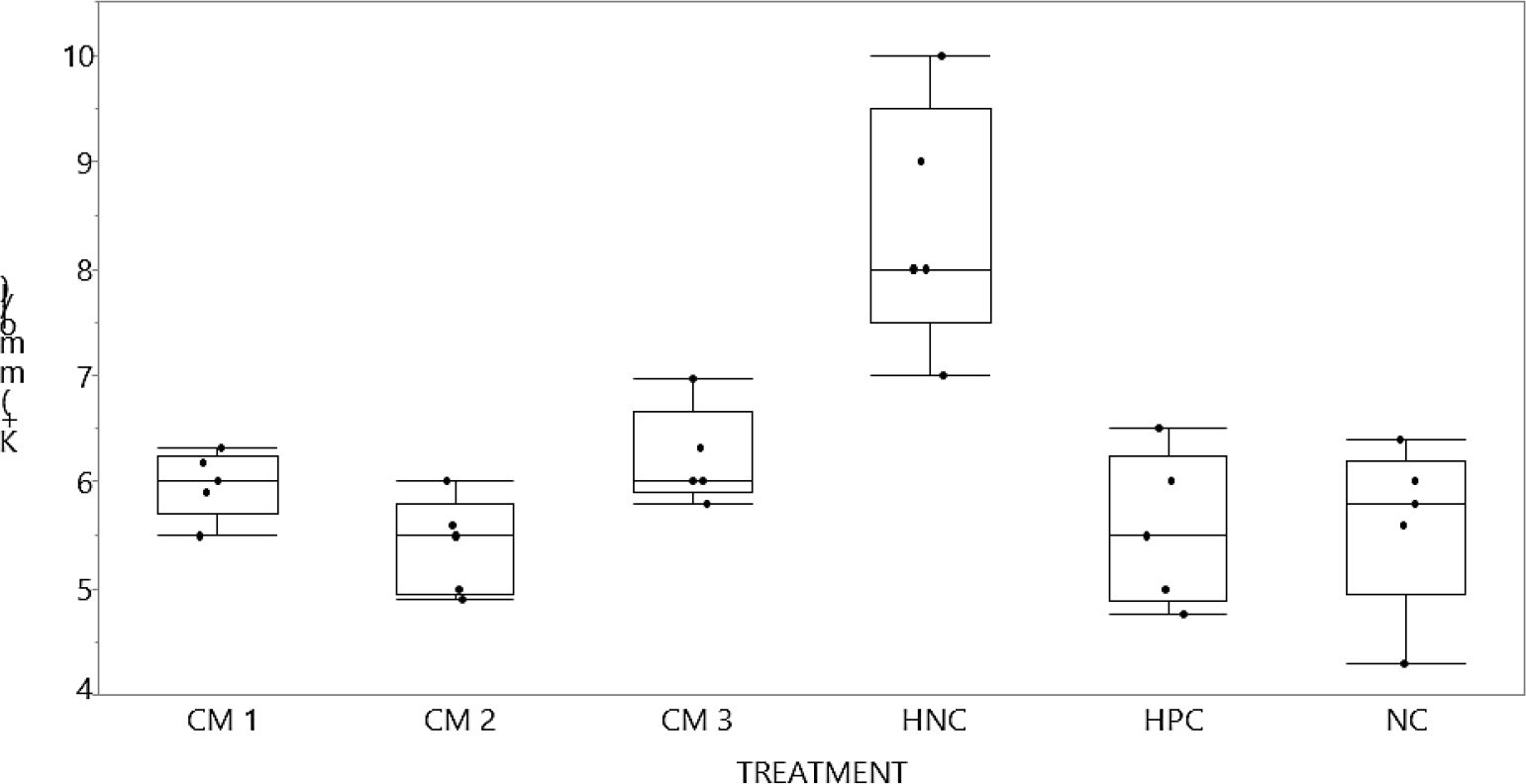
Figure 4:
Potassium ions; (K+ (mmol/l)) graph compared post treatments. Different milk treatments (CM) compared to positive control. Non-parametric comparison with positive control using Dunn Method for joint ranking. Control group:-HPC, p-value statistically significant for L-NAME only (HNC) at 0.0435* and for 1-way test chi square approximation at 0.0084*. K+:-Serum potassium ions; NC:-normal control; PC:-positive control (ref. group) amlodipine 10 mg/kg/day; HNC:- Negative control for hypertension (groups given L-NAME only [50 mg/kg/day]); L-NAME:-nitro-l-arginine methyl ester; CM1:-given raw camel milk at (100 mg/kg/day); CM3:-given raw camel milk at (300 mg/kg/day); CM5:-given raw camel milk at (500 mg/kg/day).
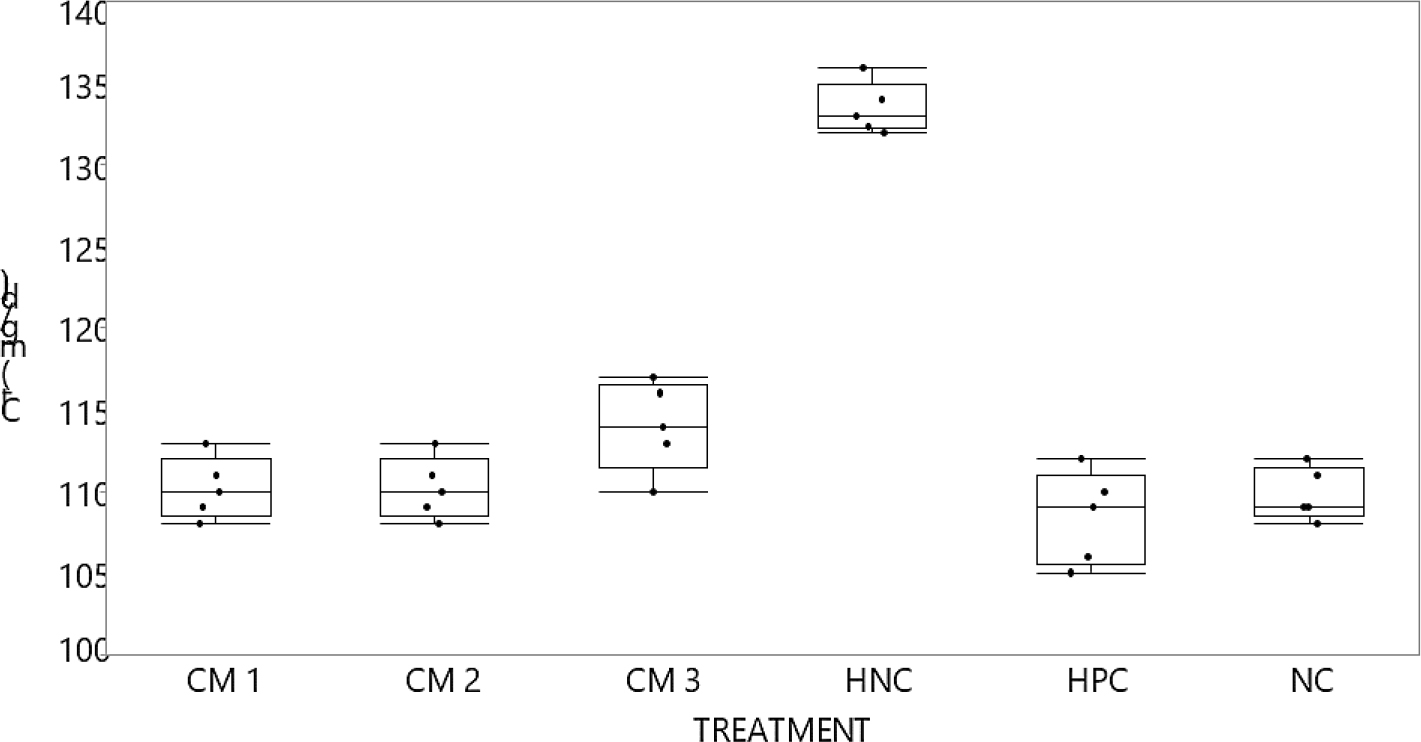
Figure 5:
Chloride ions; Cl– (mg/dL) graph compared to different interventions. Graph of Milk (CM) treatments compared to controls. Non-parametric comparison with positive control using Dunn Method for joint ranking. Control group:-HPC, p-value statistically significant for L-NAME only (HNC) at 0.0185* and for 1-way test chi square approximation at 0.0028*. Cl–:-serum chloride ions; NC:-normal control; PC:-positive control (ref. group) amlodipine 10 mg/kg/day; HNC:- Negative control for hypertension (groups given L-NAME only [50 mg/kg/day]); L-NAME:-nitro-l-arginine methyl ester; CM1:-given raw camel milk at (100 mg/kg/day); CM3:-given raw camel milk at (300 mg/kg/day); CM5:-given raw camel milk at (500 mg/kg/day).
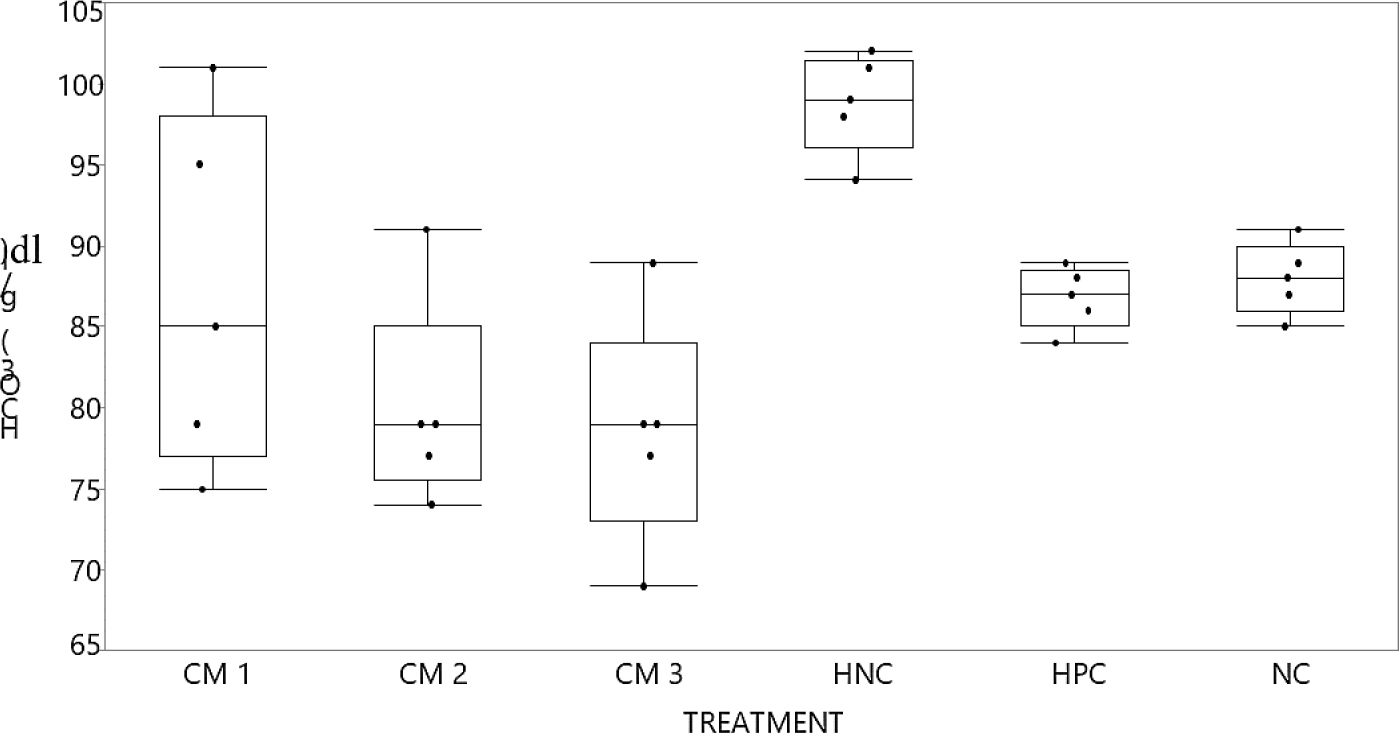
Figure 6:
Bicarbonate ions; (HCO3– (mg/dL) graph compared post interventions. Graph of Bicarbonate ions; (HCO3– (mg/dL) contrasted to controls. Non-parametric comparison with positive control using Dunn Method for joint ranking. Control group:-HPC, there is no statistical significance for p-value but there is for 1-way test chi square approximation at 0.0021*. HCO3–:-serum bicarbonate ions; NC:-normal control; PC:-positive control (ref. group) amlodipine 10 mg/kg/day; HNC:- Negative control for hypertension (groups given L-NAME only [50 mg/kg/day]); L-NAME:-nitro-l-arginine methyl ester; CM1:-given raw camel milk at (100 mg/kg/day); CM3:-given raw camel milk at (300 mg/kg/day); CM5:-given raw camel milk at (500 mg/kg/day).
Toxicity studies encompassed complete acute, sub-acute, chronic, and sub-chronic assessments of the material under study within the defined limits of what was assessed relative to its toxicity profiles. The result of findings in this present study; the examination of toxicological properties of raw camel milk orally administered by gavage via acute and sub chronic means in rats induced with hypertension has substantially agreed with (Balogun and Ashafa, 2016) earlier studies in both principle and concept. Thus, the absence of adverse consequences arising from acute studies or the zero mortality of any rat due to raw CM administration as depicted in Table 1; can all point to CM’s relative safety.
The nil death of any rat in addition to none of them showing any signs or symptoms of toxicity consequent from raw CM oral gavage administration to the studied rats was similar in both the lower and higher doses used in the acute study; that is 2000 and 5000 mg/kg in the earlier study. All the rats have survived beyond both 48 hr and cumulative 14 days of observation.
The LD50 as determined in the study above 5000 mg/kg was because the studied rats that received 5000 mg/kg CM had survived initial threshold of 2 and 14 days critical observation stages and they were also all alive until end of the study. Similarly, this found LD50 above 5000 mg/kg simply signifies that raw CM is essentially safe and not toxic. Literature regarding raw milk toxicity studies for other animals not only for camel milk is unfortunately, scarce or even none/nowhere to be found; therefore comfortable comparison cannot be easily made with findings of this study; probably because milk of almost all animals that feed their younger ones with it is generally assumed to be safe and non-toxic due to the fact that it is produced by mammary glands secretion from hormonal action.
Raw camel milk as natural product can be compared with other natural liquid fluids produced by other living organisms which appeared and are safe although some exceptions to this could be found through some plant extracts. In human nutrition; milk is being regarded as food-grade liquid of utmost importance because the health benefit effect of animal milk is incontrovertible due to its vast nutrient content (Akinyemiet al., 2021). Animals’ milk is consumed generally widely either in its raw/un-processed form or when it is prepared or converted from its natural state to another special form (as liquid, powdered, pasteurized, condensed, heat-treated or UHT-treated) medium (Becker-Algeri, 2016; Benkerroum, 2016).
Similarly, oral raw CM administration in different doses to the rats in sub chronic evaluation repeatedly for 28-days as presented in Table 2, have shown that neither were any mortality nor treatment-related sign of toxicity in all the groups at the lower 100 mg/kg or medium 300 mg/kg; but at the higher 500 mg/kg dose one rat in the group showed sluggish movement by 3rd week as highlighted. For the sub-chronic phase too, the animals’ body weights have not changed significantly between initial and final weights when controls was compared with CM treatment; although this data is not presented here.
In this research, during this sub-acute stage findings of the rats overall behavior in respect with being agile, responding to stimuli and nil absence to clinical signs associated with local or systemic toxic effects is consistent and have agreed with (Traeselet al., 2014) and (Prasanthet al., 2015) earlier findings in their separate toxicity studies.
Another area where this research finding was in agreement with another earlier (Klaassen, 2001) is with respect to unnoticeable change observed in either feed intake or water consumption. In essence this implies that there is no disruption of their carbohydrate, protein and or fat metabolisms; a milestone criteria confirming relative safety of the investigated milk. Normally vulnerability to some few possible toxic substances, may results in some increases not only in external weight and but also for internal organ weights in sync with (Carol, 1995).
The raw camel milk consumed by rats in this study may not have exerted any toxic effect on their pair of kidneys that handles any food or ingested foreign material metabolism and its biotransformation; confirming renal organ integrity or its functions being intact. Renal values found for the CM treatment were all within normal reference range and have not deviated significantly with the figures obtained for positive and the normal instituted controls.
Creatinine and urea as 2 waste products of protein metabolism conveyed through kidneys and filtered as urine are therefore important metrics of kidney organ integrity.
CONCLUSION
In conclusion the results of this study based on the findings from both acute and sub-chronic raw CM administration to rats have implied that it can be regarded as relatively safe and non-toxic because no any rat death was caused by it in the studies and neither was any significant abnormality. Further research is required in future with regards to fully elucidating various medical potentials of raw CM in view of its several acclaimed therapeutic importance.
References
- Agrawal R. P., Budania S., Sharma P., Gupta R., Kochar D. K., Panwar R. B., Sahani M. S., et al. (2007) Zero prevalence of diabetes in camel milk consuming Raica community of northwest Rajasthan, India. Diabetes Research and Clinical Practice 76: 290-296 https://doi.org/10.1016/j.diabres.2006.09.036 | Google Scholar
- Akinyemi M. O., Ayeni K. I., Ogunremi O. R., Adeleke R. A., Oguntoyinbo F. A., Warth B., Ezekiel C. N., et al. (2021) A review of microbes and chemical contaminants in dairy products in sub-Saharan Africa. In Comprehensive Reviews in Food Science and Food Safety 20: 1188-1220 https://doi.org/10.1111/1541-4337.12712 | Google Scholar
- Akinyemi M. O., Braun D., Windisch P., Warth B., Ezekiel C. N.. (2022) Assessment of multiple mycotoxins in raw milk of three different animal species in Nigeria. Food Control 131: Article 108258 https://doi.org/10.1016/j.foodcont.2021.108258 | Google Scholar
- Alebie G., Yohannes S., Worku A.. (2017) Therapeutic applications of camel’s milk and urine against cancer: Current development efforts and future perspectives. Journal of Cancer Science and Therapy 9: 468-478 https://doi.org/10.4172/1948-5956.1000461 | Google Scholar
- Balogun F. O., Tom Ashafa A. O. T.. (Array) Acute and sub-chronic oral toxicity evaluation of aqueous root extract of Sond. in Wistar rats. Evidence-Based Complementary and Alternative Medicine: eCAM : Article ID 3509323 https://doi.org/10.1155/2016/3509323 | Google Scholar
- Becker-Algeri T. A., Castagnaro D., de Bortoli K., de Souza C., Drunkler D. A., Badiale-Furlong E., et al. (2016) Mycotoxins in bovine milk and dairy products: A review. Journal of Food Science 81: R544-R552 https://doi.org/10.1111/1750-3841.13204 | Google Scholar
- Benkerroum N.. (2016) Mycotoxins in dairy products: A review. International Dairy Journal 62: 63-75 https://doi.org/10.1016/j.idairyj.2016.07.002 | Google Scholar
- Carol S. A.. (1995) Acute, Sub chronic and Chronic Toxicology. https://doi.org/10.1016/j.idairyj.2016.07.002 | Google Scholar
- (2018) Centre for Food Security and Public Health in association with Institute for International Cooperation in Animal Biologics : 2017-2018 https://doi.org/10.1016/j.idairyj.2016.07.002 | Google Scholar
- Desouky M. M., Salama H. H.. (2021) Preparation and properties of children food after weaning using camels’ milk and guadar cereal nanoparticles. Journal of Food Processing and Preservation 45: Article e15012 https://doi.org/10.1111/jfpp.15012 | Google Scholar
- Desouky M. M., Salama H. H., El-Sayed S. M.. (2019) The effects of camel milk powder on the stability and quality properties of processed cheese sauce. Acta Scientiarum Polonorum. Technologia Alimentaria 18: 349-359 https://doi.org/10.17306/J.AFS.0645 | Google Scholar
- Dogondaji R., Lawal M., Wasagu R., Abubakar I., Umar S.. (2024) Hepatoprotective effect of camel () raw milk on some increased liver biomarkers in hypertensive rats. J. Innov. Res. Life Sci. 6: 23-28 https://doi.org/10.17306/J.AFS.0645 | Google Scholar
- Dogondaji R. A., Lawal M., Wasagu R. S. U., Yakubu A.. (2023)) [Research report]. Camel () raw milk’s hypotensive roles within chemical induced hypertension model in rats. Mediterranean Journal of Nutrition and Metabolism 16: 71-87 https://doi.org/10.3233/MNM-220099 | Google Scholar
- Dogondaji R. A., Wasagu R. S. U., Ismaila M. S., Abubakar K., Umar S.. (2023)a.) Raw milk proximate composition analyses for some camels () in Sokoto, Nigeria. PSM Biol. Res. 8: 62-68 https://doi.org/10.3233/MNM-220099 | Google Scholar
- Dubey U. S., Lal M., Mittal A., Kapur S.. (2016) Therapeutic potential of camel milk. Emirates Journal of Food and Agriculture 28: 164-176 https://doi.org/10.9755/ejfa.2015-04-122 | Google Scholar
- Hernández-Ledesma B., del Mar Contreras M., Recio I.. (2011) Antihypertensive peptides: Production, bioavailability and incorporation into foods. Advances in Colloid and Interface Science 165: 23-35 https://doi.org/10.1016/j.cis.2010.11.001 | Google Scholar
- Ho T. M., Zou Z., Bansal N.. (2022) Camel milk: A review of its nutritional value, heat stability, and potential food products. Food Research International 153: Article 110870 https://doi.org/10.1016/j.foodres.2021.110870 | Google Scholar
- Hussain I., Anwar J.. (2008) A study on contamination of aflatoxin M1 in raw milk in the Punjab Province of Pakistan. Food Control 19: 393-395 https://doi.org/10.1016/j.foodcont.2007.04.019 | Google Scholar
- (2012) https://doi.org/10.1016/j.foodcont.2007.04.019 | Google Scholar
- . (2021) Global industry trends, share, size, growth, opportunity and forecast 2021-2026. https://doi.org/10.1016/j.foodcont.2007.04.019 | Google Scholar
- JD. Mannfred A.H.. In CRC handbook of toxicology : 51-104 https://doi.org/10.1016/j.foodcont.2007.04.019 | Google Scholar
- Kamal-Eldin A., Ayyash M., Sobti B., Nagy P.. (2022) Camel milk : 504-513 https://doi.org/10.1016/B978-0-12-818766-1.00327-5 | Google Scholar
- Klaassen C. D.. (2001) Principles of toxicology. In Casarett and Doull’s toxicology: The basic science of poisons : 13 https://doi.org/10.1016/B978-0-12-818766-1.00327-5 | Google Scholar
- Kroll M. H., Roach N. A., Poe B., Elin R. J.. (1987) Mechanism of interference with the Jaffé reaction for creatinine. Clinical Chemistry 33: 1129-1132 https://doi.org/10.1093/clinchem/33.7.1129 | Google Scholar
- Mihic T., Rainkie D., Wilby K. J., Pawluk S. A.. (2016) The therapeutic effects of camel milk: A systematic review of animal and human trials. Journal of Evidence-Based Complementary and Alternative Medicine 21: NP110-NP126 https://doi.org/10.1177/2156587216658846 | Google Scholar
- Nagy P., Wernery U., Burger P., Juhasz J., Faye B.. (2021) The impact of COVID-19 on Old World Camelids and their potential role to combat a human pandemic. Animal Frontiers: The Review Magazine of Animal Agriculture 11: 60-66 https://doi.org/10.1093/af/vfaa048 | Google Scholar
- Omar S. S.. (2016) Aflatoxin M1 levels in raw milk, pasteurized milk and infant formula. Italian Journal of Food Safety 5: 5788 https://doi.org/10.4081/ijfs.2016.5788 | Google Scholar
- (1995) Guideline for testing of chemicals. Repeated dose 28-day oral toxicity study in rodents. https://doi.org/10.4081/ijfs.2016.5788 | Google Scholar
- (2008) Guideline for the testing of chemicals Test No. 407: Repeated dose 28-day oral toxicity study in rodents. https://doi.org/10.4081/ijfs.2016.5788 | Google Scholar
- Prandini A., Tansini G., Sigolo S., Filippi L., Laporta M., Piva G., et al. (2009) On the occurrence of aflatoxin M1 in milk and dairy products. Food and Chemical Toxicology 47: 984-991 https://doi.org/10.1016/j.fct.2007.10.005 | Google Scholar
- Prasanth K. M., Suba V., Ramireddy B., Srinivasa B. P.. (2015) Acute and Sub chronic Oral Toxicity Assessment of the ethanolic Extract of the root of (Flacourtiaceae) in Rodents. Tropical Journal of Pharmaceutical Research 14: 1849-1855 https://doi.org/10.4314/tjpr.v14i10.16 | Google Scholar
- Raza M., Al-Shabanah O. A., El-Hadiyah T. M., Al-Majed A. A.. (2002) Effect of prolonged vigabatrin treatment on hematological and biochemical parameters in plasma, liver and kidney of Swiss albino mice. Sci. of Pharmacog. 70: 135-145 https://doi.org/10.4314/tjpr.v14i10.16 | Google Scholar
- Shakeel K., Rabail R., Iahtisham-Ul-Haq S., Sehar S., Nawaz A., Manzoor M. F., Walayat N., Socol C. T., Maerescu C. M., Aadil R. M., et al. (2022) Camel milk protectiveness toward multiple liver disorders: A review. Frontiers in Nutrition 9: Article 944842 https://doi.org/10.3389/fnut.2022.944842 | Google Scholar
- Soleymanzadeh N., Mirdamadi S., Mirzaei M., Kianirad M.. (2019) Novel b-casein derived antioxidant and ACE-inhibitory active peptide from camel milk fermented by Leuconostoc lactis PTCC1899: Identification and molecular docking. International Dairy Journal 97: 201-208 https://doi.org/10.1016/j.idairyj.2019.05.012 | Google Scholar
- Teo S. D., Stirling S., Thomas A., Kiorpes A., Vikram K.. (2002) A 90- day oral gavage toxicity study of D-methylphenidate and D, L methylphenidate in Sprague-Dawley rats. The Toxicologist 179: 183-196 https://doi.org/10.1016/j.idairyj.2019.05.012 | Google Scholar
- Traesel G. K., de Souza J. C., de Barros A. L., Souza M. A., Schmitz W. O., Muzzi R. M., Oesterreich S. A., Arena A. C., et al. (2014) Acute and sub-acute (28 days) oral toxicity assessment of the oil extracted from Acrocomia aculeata pulp in rats. Food and Chemical Toxicology 74: 320-325 https://doi.org/10.1016/j.fct.2014.10.026 | Google Scholar
- Uriyo A. P., Singh B. R.. (1974) Practical soil chemistry manual : 12-14 https://doi.org/10.1016/j.fct.2014.10.026 | Google Scholar
- van de Wiel M. A.. (2002) Exact distributions of multiple comparisons rank statistics. Journal of the American Statistical Association 97: 1081-1089 https://doi.org/10.1198/016214502388618898 | Google Scholar
- Wills M. R., Savory J. O.. (1981) Biochemistry of renal failure. Annals of Clinical and Laboratory Science 11: 292-299 https://doi.org/10.1198/016214502388618898 | Google Scholar
- Wybenga D. R. D., Glorgia J., Pileggi V. J.. (1971) Determination of serum urea by diacetyl monoxime method. J. Clin. Chem. 17: 891-898 https://doi.org/10.1198/016214502388618898 | Google Scholar

