ABSTRACT
Introduction
the current epidemic of COVID-19 requires a lot of efforts to be controlled involving pharmacotherapy and clinical pharmacology studies. Aim: to introduce a brief review of drugs under investigation for COVID-19.
Methodology
Our project is a systematic review of many medications being studied for their effectiveness and safety in COVID-19 management.
Results
Antiviral drugs (osletmivir, ramdisvir), corticosteroids (dexamethasone), immunomodulators (anakinera, toculizumab), antithrombotic (antiplatletes, anticoagulants) are still under investigation although preliminary results are promising.
Conclusion
Yet, few medications have been approved by FDA for management of COVID-19 but drugs have been used in clinical trials under FDA emergency use authorization. Ongoing clinical trials are essential to establish their long-term safety and effectiveness. Key points: 1. Scientists are testing different medicines, such as antivirals, steroids, immune-boosting drugs, and blood thinners, to see if they can help treat COVID-19. 2. Some of these drugs show early promise, but only a few have official approval for use. 3. More research and clinical trials are needed to make sure these treatments are safe and work well in the long run.
INTRODUCTION
Coronaviruses (CoVs) are a family of RNA viruses classified under the subfamily Orthocoronavirinae, within the Coronaviridae family and the order Nidovirales. This group includes alpha-, beta-, gamma-, and delta-coronaviruses. Notably, beta-coronaviruses have been responsible for three major outbreaks in the past two decades: Severe Acute Respiratory Syndrome (SARS-CoV) in 2002, Middle East Respiratory Syndrome (MERS-CoV) in 2012, and the ongoing COVID-19 pandemic caused by SARS-CoV-2, first identified in late 2019 (Banerjeeet al., 2019; The Lancet Oncology, 2020).
As of July 31, 2020, the World Health Organization (WHO) had received extensive global reporting on COVID-19 cases (Figure 1; World Health Organization, 2020). These data underscore the importance of robust clinical and pharmacological responses to curb the pandemic.
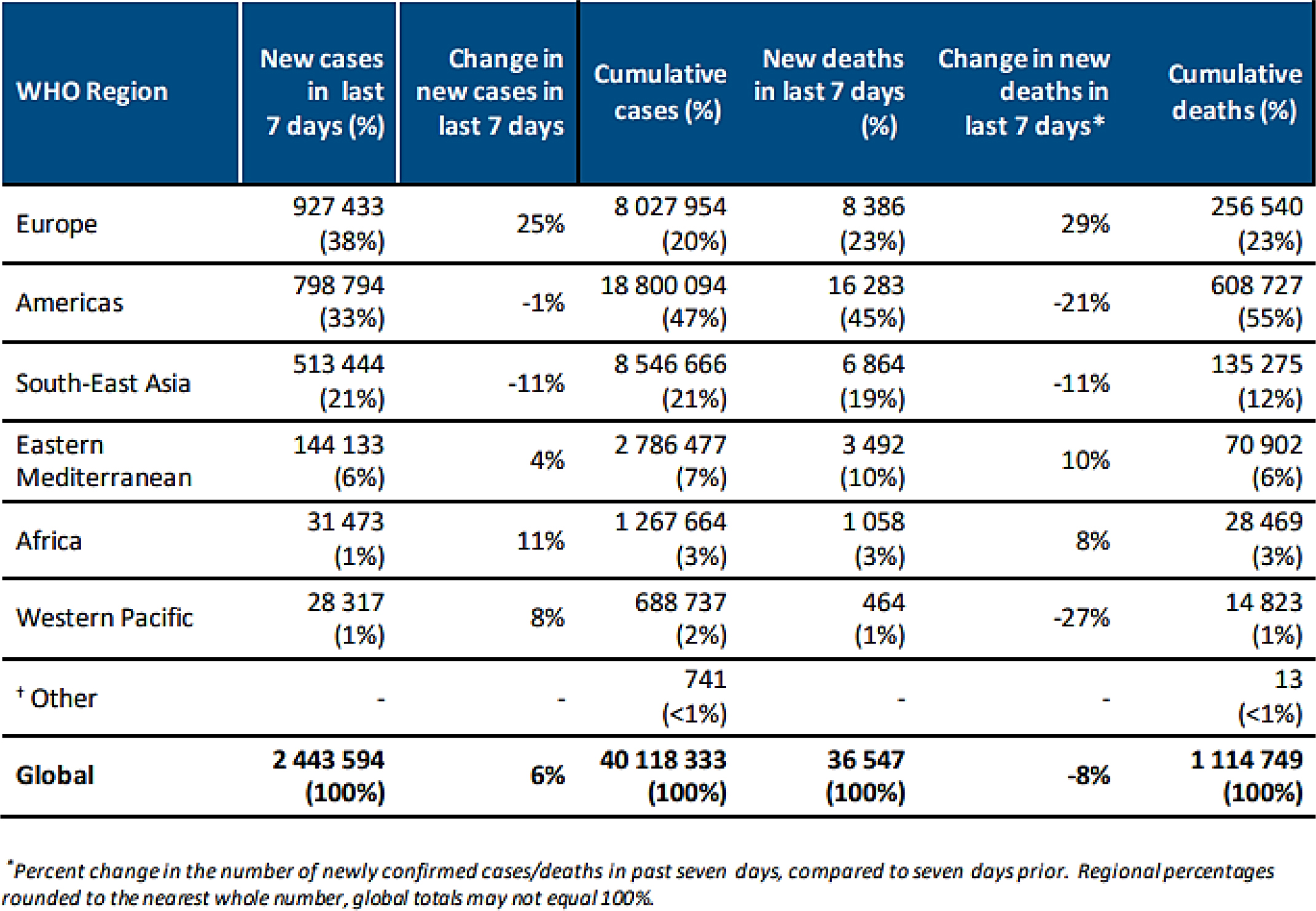
Figure 1:
COVID-19 Situation in numbers 20/10/2020 (by WHO Region) (Al-Tawfiqet al., 2014).
Objectives
To introduce a brief review of medications under investigation for COVID-19 management showing their efficacy and safety with evidence by clinical trials
METHODOLOGY
Our study is a systematic review of many medications being studied for their effectiveness and safety in COVID-19 management. We used PubMed search tool using term “management of COVID-19 by pharmacotherapy” resulting in many clinical trials and few systematic review.
Literature Review
Nutritional interventions
Nutritional Supportive Therapies
Nutritional approaches have gained attention for their potential to modulate immune responses in patients with COVID-19. Vitamins and minerals such as vitamin C, D, and zinc may help reinforce the host defense mechanisms against viral infections. These agents play a supportive role in recovery and are commonly recommended as adjunctive therapy, especially in cases involving immune dysregulation (Figure 2; Zhang and Liu, 2020).

Figure 2:
General supportive treatments of COVID-19 (Amiciet al., 2006).
Coronavirus‐specific treatments
Chloroquine/Hydroxychloroquine
These antimalarial compounds initially showed promise due to their ability to interfere with viral entry via the ACE2 receptor. However, data from clinical trials have yielded inconsistent efficacy and raised concerns over cardiac safety, particularly QT interval prolongation, limiting their use to experimental settings (Savarinoet al., 2003; Vincentet al., 2005; Fanget al., 2020; Borbaet al., 2020; CredibleMeds, 2020; Rosa and Santos, 2020; Chorinet al., 2020).
Promazine
As an older antipsychotic, promazine has demonstrated potential antiviral activity by disrupting the viral entry process, but it has not progressed beyond preclinical evaluation (Zhang and Yap, 2004).
Antiviral treatments
Summary of antiviral agents is shown in Figures 5–7.

Figure 5:
Antiviral drugs for COVID-19.
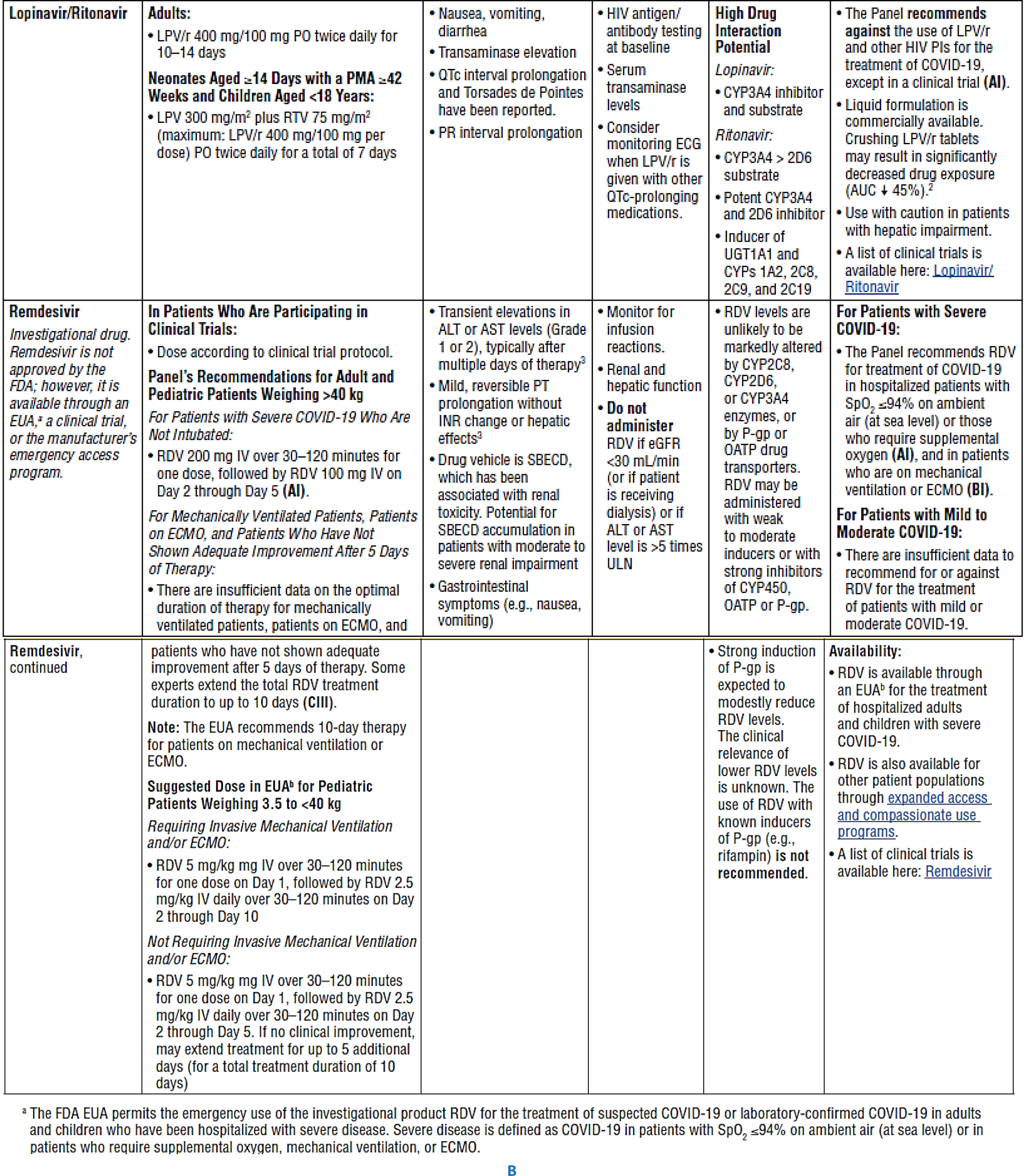
Figure 6:
Characteristics of Potential Antiviral Agents Under Evaluation for COVID-19 (Food and Drug Administration, 2020).
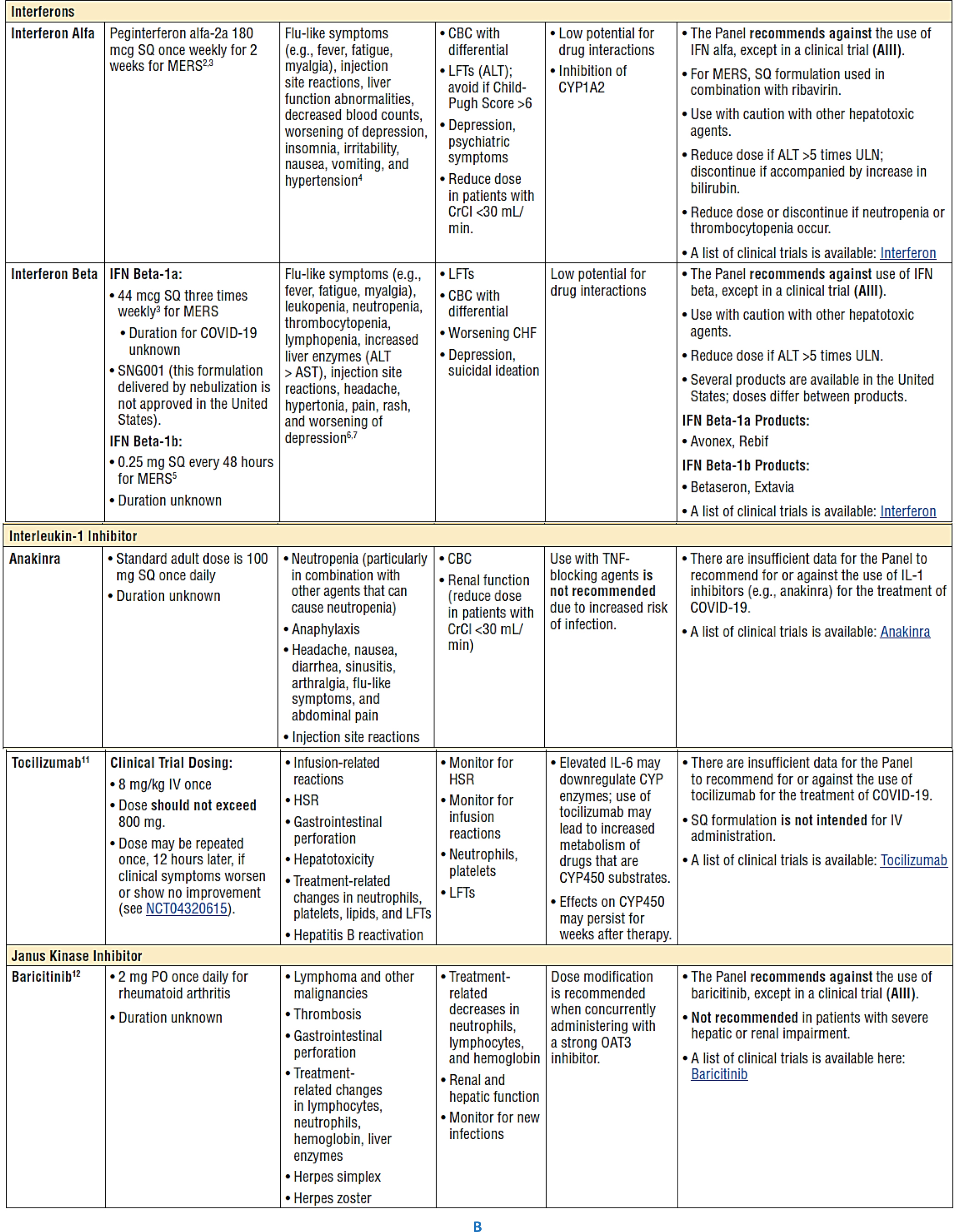
Figure 7:
Characteristics of Immune-Based Therapy Under Evaluation for Treatment of COVID-19 (Food and Drug Administration, 2020).
Lopinavir/ritonavir (Kaletra)
This protease inhibitor combination, used in HIV management, showed modest efficacy in early coronavirus outbreaks. Its use in COVID-19 has been discouraged outside clinical trials due to pharmacokinetic challenges and limited clinical improvement (Tsang and Zhong, 2003; Kimet al., 2016; Caoet al., 2020).
Remdesivir
A nucleotide analog with broad antiviral properties, remdesivir has demonstrated inhibition of coronavirus replication in animal and human studies. It is frequently administered to hospitalized patients requiring respiratory support, typically in a 5- to 10-day course depending on clinical status (Agostiniet al., 2018; Sheahanet al., 2020; Beigelet al., 2020; Goldmanet al., 2020).
Nelfinavir
Azithromycin
Niclosamide and Ivermectin
Oseltamivir
Primarily used for influenza, oseltamivir has been included in some investigational combinations against COVID-19, although its individual efficacy remains unclear (Rosa and Santos, 2020).
Immune-Based Therapy Under Evaluation for Treatment of COVID-19
Convalescent Plasma and Immune Globulins
Interleukin-1 Inhibitors
Interleukin-6 Inhibitors
Agents such as tocilizumab, sarilumab, and siltuximab target IL-6 pathways involved in cytokine release syndromes. Despite their theoretical benefits, current evidence is inadequate to suggest routine use in COVID-19 (Press Release, 2020; Sciasciaet al., 2020).
Interferons (Alfa, Beta)
Janus Kinase Inhibitors (e.g., Baricitinib)
JAK inhibitors, including baricitinib, are not currently recommended for general COVID-19 treatment and should only be administered under controlled clinical research (Richardsonet al., 2020).
Antithrombotic Therapy in Patients with COVID-19
Chronic Anticoagulant and Antiplatelet Therapy
Patients already prescribed antithrombotic therapy for other conditions should continue their regimen upon COVID-19 diagnosis (AIII).
Venous Thromboembolism Prophylaxis and Screening
Non-hospitalized individuals with COVID-19 should not receive anticoagulation prophylaxis unless another indication exists (AIII). Hospitalized patients should receive standard VTE prophylaxis, but routine post-discharge prophylaxis is not recommended unless specific high-risk factors apply (BI).
Screening and Management
Routine screening for thrombotic events in asymptomatic COVID-19 patients is not supported by current evidence (BIII). However, if there is rapid clinical deterioration suggestive of thromboembolism, further evaluation and management are warranted (AIII).
Treatment
In cases of confirmed or highly suspected thromboembolism where imaging is not feasible, standard anticoagulant therapy should be initiated (AIII). Patients undergoing ECMO, CRRT, or catheter-based interventions should also receive antithrombotic therapy as per institutional protocols (AIII).
Selection of Anticoagulant or Antiplatelet Drugs for Patients with COVID-19
For hospitalized patients, low molecular weight or unfractionated heparin is preferred due to better control and fewer interactions (AIII). Outpatients on warfarin with limited INR access may transition to direct oral anticoagulants unless contraindicated (American Society of Hematology, 2020).
Other compounds
α‐Lipoic acid (ALA)
ALA, long used for liver and nerve-related disorders, exhibits strong antioxidant properties by enhancing intracellular glutathione and neutralizing oxidative stress. It may mitigate viral infectivity by counteracting oxidative damage and G6PD-related vulnerabilities to coronaviruses (Sachse and Willms, 1980; Tibulloet al., 2017; El-Senouseyet al., 2018; Wuet al., 2008).
Dexamethasone
As a potent corticosteroid, dexamethasone has shown benefit in reducing inflammation and improving outcomes in patients with severe COVID-19. The RECOVERY trial demonstrated improved survival among ventilated patients or those needing supplemental oxygen (Horbyet al., 2020). Dexamethasone is not advised in cases without oxygen support (AI) (Molinaet al., 2020). Chronic corticosteroid users for underlying conditions should continue therapy, possibly with stress-dose adjustments (AIII) (Kaiseret al., 2020).
Empiric Broad-Spectrum Antimicrobial Therapy
In patients with COVID-19 and intense or vital illness, there are inadequate records to endorse empiric broad-spectrum antimicrobial therapy in the absence of any other indication (BIII).
If antimicrobials are initiated, the Panel recommends that their use must be reassessed every day so that you can decrease the unfavorable effects of needless antimicrobial therapy (AIII).
Considerations for Certain Concomitant Medications in Patients with COVID-19
Angiotensin-Converting Enzyme (ACE) Inhibitors and Angiotensin Receptor Blockers (ARBs)
Persons with COVID-19 who are taking NSAIDs for a comorbid condition should continue therapy as previously directed by their physician (AIII). The Panel recommends that there be no distinction in the use of antipyretic strategies (e.g., with acetaminophen or NSAIDs) among patients with or without COVID-19 (AIII; Patel and Verma, 2020; American College of Cardiology, 2020).
Ibuprofen
Ibuprofen, a Nonsteroidal Anti-Inflammatory Drug (NSAID), is an activator of ACE2 receptors, similar to ACE inhibitors or ARBs. Their utilization can result in an increased hazard of contracting COVID-19 (Fanget al., 2020). Since fatal lung failure triggered through SARS-CoV infections can be managed through blocking the renin-angiotensin pathway (Kubaet al., 2005), ibuprofen might not be harmful. However, there is no strong evidence suggesting a link between NSAID use and worsening symptoms due to SARS-CoV-2 infection.
Indomethacin
Amiciet al., (2006) demonstrated that indomethacin, a widely used NSAID and a potential Cyclooxygenase (COX) inhibitor, exhibits antiviral activity against SARS-CoV. Remarkable inhibition of SARS-CoV-infected cells by more than 99% at non-toxic concentrations was also observed. This suggests probable efficacy of indomethacin against SARS-CoV-2.
RESULTS
Nutritional intervention: Vitamins, minerals, and immunomodulators play an important role in enhancing the body’s immunity against several viruses, including SARS-CoV (Zhang and Liu, 2020). Chloroquine and hydroxychloroquine are potent inhibitors of SARS coronavirus infection but are not recommended for use outside clinical trials (Savarinoet al., 2003).
Currently, many trials are testing the effect of azithromycin in conjunction with hydroxychloroquine on the course of disease in people with SARS-CoV-2, showing its efficacy (Gautretet al., 2020; Molinaet al., 2020). However, the azithromycin and hydroxychloroquine combination is not recommended for use outside clinical trials due to potential interactions (e.g., prolonged QT-interval) (Borbaet al., 2020; CredibleMeds, 2020; Rosa and Santos, 2020).
Although lopinavir/ritonavir showed potential during the early disease phase in conjunction with interferons, studies have since shown limited benefit in COVID-19 patients due to poor pharmacokinetics (Caoet al., 2020).
Remdesivir has shown efficacy in preclinical studies by reducing viral load and improving lung function. It is currently recommended for hospitalized patients requiring oxygen or mechanical support (Agostiniet al., 2018; Sheahanet al., 2020; Beigelet al., 2020).
Nelfinavir has also demonstrated significant inhibitory effects on SARS-CoV replication and may be a candidate for further COVID-19 trials (Yamamotoet al., 2004). Oseltamivir, while effective against influenza, is being studied in combination with other drugs for potential use against SARS-CoV-2 (Rosa and Santos, 2020).
Niclosamide and ivermectin both show strong in vitro antiviral properties, with ivermectin reducing viral RNA by thousands-fold within 48 hr (Calyet al., 2020; Wuet al., 2004).
ALA acts as an antioxidant, potentially reducing oxidative stress and related complications during viral infection, including COVID-19 (Tibulloet al., 2017; Wuet al., 2008).
Dexamethasone has proven clinical benefit in patients requiring respiratory support by dampening systemic inflammation (Horbyet al., 2020).
Use of broad-spectrum antibiotics is discouraged unless secondary infection is suspected. Convalescent plasma, IL-1/IL-6 inhibitors, interferons, and baricitinib remain under investigation and are not yet recommended for routine care (Ahnet al., 2020; Shakooryet al., 2016; Cavalliet al., 2020; Sciasciaet al., 2020; Al-Tawfiqet al., 2014; Richardsonet al., 2020).
Convalescent Plasma and Immune Globulins
Antithrombotic Therapy
Antithrombotic interventions, including heparins and oral anticoagulants, are used only when clinically indicated. Choice of drug depends on the patient’s condition and setting, with LMWH and UFH preferred in hospitalized patients (American Society of Hematology, 2020).
Anakinra
Tocilizumab
There are insufficient data to recommend either for or against the use of interleukin-6 (IL-6) inhibitors (tocilizumab) for the treatment of COVID-19 (Press Release, 2020; Sciasciaet al., 2020).
Interferons
Baricitinib
It is not recommended to use Janus kinase (JAK) inhibitors (e.g., baricitinib) for the treatment of COVID-19, except in the context of a clinical trial (Richardsonet al., 2020).
Antithrombotic Therapy
Patients with COVID-19 who experience an incident thromboembolic event or who are highly suspected to have thromboembolic disease at a time when imaging is not possible should be managed with therapeutic doses of anticoagulant therapy as per the standard of care for patients without COVID-19. Patients with COVID-19 who require extracorporeal membrane oxygenation or continuous renal replacement therapy or who have thrombosis of catheters or extracorporeal filters should be treated with antithrombotic therapy per institutional protocols. Low molecular weight heparin or unfractionated heparin may be preferred in hospitalized, critically ill patients because of their shorter half-lives, ability to be administered intravenously or subcutaneously, and fewer drug-drug interactions compared with oral anticoagulants. Outpatients receiving warfarin who are unable to get international normalized ratio monitoring during isolation may be candidates for direct oral anticoagulant therapy. Patients with mechanical heart valves, ventricular assist devices, valvular atrial fibrillation, or antiphospholipid antibody syndrome or patients who are lactating should continue treatment with warfarin therapy (American Society of Hematology, 2020).
Angiotensin-Converting Enzyme (ACE) Inhibitors and Angiotensin Receptor Blockers (ARBs)
It is not recommended to use ACE inhibitors or ARBs for the treatment of COVID-19 outside the setting of a clinical trial (Patel and Verma, 2020).
HMG-CoA Reductase Inhibitors (Statins)
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs)
There should be no difference in the use of antipyretic strategies (e.g., with acetaminophen or NSAIDs) between patients with or without COVID-19 (Food and Drug Administration, 2020; Bancoset al., 2009).
Ibuprofen is an activator of ACE2 receptors; its usage can lead to increased risk of contracting COVID-19 (Fanget al., 2020).
Indomethacin
Indomethacin exhibits antiviral activity against SARS-CoV. Remarkable inhibition against SARS-CoV-infected Vero cells by more than 99% at concentrations that were non-toxic for uninfected cells has also been observed. This suggests probable efficacy of indomethacin against SARS-CoV-2 (Amiciet al., 2006).
DISCUSSION
As shown in our results, clinical trials are still in progress and until now there are no definitive results for the efficacy of studied medications for management of COVID-19. Although preliminary findings for some medications are promising, others show that harms may outweigh benefits.
Nutritional interventions should be initiated as early as diagnosis is suspected to enhance patient immunity against respiratory viruses and any opportunistic bacterial respiratory infections (Zhang and Liu, 2020).
Chloroquine, hydroxychloroquine, lopinavir/ritonavir, anakinra, tocilizumab, interferons, baricitinib, ACE inhibitors, ARBs, and statins are still not recommended for COVID-19 management except in clinical trials (Vincentet al., 2005; Caoet al., 2020; Aoubaet al., 2020; Cavalliet al., 2020; Sciasciaet al., 2020; Al-Tawfiqet al., 2014; Arabiet al., 2019; Richardsonet al., 2020; Patel and Verma, 2020; Fedsonet al., 2020).
Remdesivir, nelfinavir, azithromycin, niclosamide, ivermectin, oseltamivir, indomethacin, convalescent plasma, and immune globulins have shown antiviral activity against SARS-CoV-2, though clinical trials are still in progress (Rosa and Santos, 2020; Beigelet al., 2020; Calyet al., 2020; Wuet al., 2004; Amiciet al., 2006; Ahnet al., 2020; Yeet al., 2020).
Dexamethasone has shown improvement in COVID-19 patients who developed Systemic Inflammatory Response Syndrome (SIRS) and pulmonary inflammation (Horbyet al., 2020).
ALA, due to its antioxidant effects, may help mitigate complications linked to viral oxidative stress (Tibulloet al., 2017; Wuet al., 2008).
Antithrombotic therapy is not used unless there is a clear indication for either prophylaxis or treatment. Low Molecular Weight Heparin (LMWH) or Unfractionated Heparin (UFH) are favored for critically ill patients, while Direct Oral Anticoagulants (DOACs) or warfarin are appropriate for stable outpatients when monitoring is feasible (American Society of Hematology, 2020).
For hospitalized, critically ill patients, LMWH or UFH may be preferred because of their shorter half-lives, intravenous or subcutaneous administration, and fewer drug-drug interactions compared with oral anticoagulants. For outpatients, warfarin or DOACs are preferred. In cases where international normalized ratio monitoring is not possible during isolation, DOAC therapy is recommended. Patients with mechanical heart valves, ventricular assist devices, valvular atrial fibrillation, or antiphospholipid antibody syndrome or lactating patients should continue warfarin therapy (American Society of Hematology, 2020).
CONCLUSION
Finally, treating physicians should consider patients’ co-morbidities and efficacy of medications used in patient management. Cardiac patients taking antiarrhythmic drugs should avoid medications causing QT-interval prolongation (azithromycin, chloroquine and hydroxychloroquine). Corticosteroids using in hypertensive and diabetic patients require monitoring BP and RBG. Nutritional interventions (ex: vitamin C and zinc) have important role in enhanching immunity against viral infection on long term use.
Medications that show antiviral activity against COVID-19, have proven benefits in SARS-COV2 management (remedisvir, nilfenvir, oseltamvir, azithromycin, ivermectin, niclosamid). Corticosteroids improve SIRS and respiratory symptoms associated with COVID-19 infection.
ALA has antioxidant activity and prevents oxidative stress and neuropathy associated with viral infection.
Anti-thrombotic therapy is used only when there is an indication for its use whether treatment or prophylaxis. Injectable anticoagulants are preferred in hospitalized patients while warfarin and direct oral anticoagulants are preferred in outpatients.
Other medications still do not show benefits in COVID-19 management and their use is limited to clinical trials (Chloroquine, hydroxychloroquine, Lopinavir/ritonavir, anakinra, tocilizumab, Interferons, Baricitinib, ACEIs, ARBs and statins).
References
- Ahn J. Y., Sohn Y., Lee S. H.. (2020) Use of convalescent plasma therapy in two COVID-19 patients with acute respiratory distress syndrome in Korea. Journal of Korean Medical Science 35: e149 https://doi.org/10.3346/jkms.2020.35.e149 | Google Scholar
- Agostini M. L., Andres E. L., Sims A. C., Graham R. L., Sheahan T. P., Lu X., et al. (2018) Coronavirus susceptibility to the antiviral remdesivir (GS-5734) is mediated by the viral polymerase and the proofreading exoribonuclease. mBio 9: e00221-18 https://doi.org/10.1128/mBio.00221-18 | Google Scholar
- Al-Tawfiq J. A., Momattin H., Dib J., Memish Z. A.. (2014) Ribavirin and interferon therapy in patients infected with the Middle East respiratory syndrome coronavirus: An observational study. International Journal of Infectious Diseases 20: 42-46 https://doi.org/10.1016/j.ijid.2013.12.003 | Google Scholar
- Amici C., Di Caro A., Ciucci A., Chiappa L., Castilletti C., Martella V., et al. (2006) Indomethacin has a potent antiviral activity against SARS coronavirus. Antiviral Therapy 11: 1021-1030 https://doi.org/10.1016/j.ijid.2013.12.003 | Google Scholar
- American College of Cardiology.. 2020 HFSA/ACC/AHA statement addresses concerns re: using RAAS antagonists in COVID-19.
- American Society of Hematology.. 2020 COVID-19 and VTE/anticoagulation: Frequently asked questions.
- Aouba A., Baldolli A., Geffray L.. (2020) Targeting the inflammatory cascade with anakinra in moderate to severe COVID-19 pneumonia: Case series. Annals of the Rheumatic Diseases 79: 1381-1382 https://doi.org/10.1136/annrheumdis-2020-217706 | Google Scholar
- Arabi Y. M., Shalhoub S., Mandourah Y.. (2019) Ribavirin and interferon therapy for critically ill patients with Middle East respiratory syndrome: A multicenter observational study. Clinical Infectious Diseases 70: 1837-1844 https://doi.org/10.1093/cid/ciz544 | Google Scholar
- Banerjee A., Kulcsar K., Misra V., Frieman M., Mossman K.. (2019) Bats and coronaviruses. Viruses 11: 41 https://doi.org/10.3390/v11010041 | Google Scholar
- Bancos S., Bernard M. P., Topham D. J., Phipps R. P.. (2009) Ibuprofen and other widely used non-steroidal anti-inflammatory drugs inhibit antibody production in human cells. Cellular Immunology 258: 18-28 https://doi.org/10.1016/j.cellimm.2009.03.008 | Google Scholar
- Beigel J. H., Tomashek K. M., Dodd L. E., Mehta A. K., Zingman B. S., Kalil A. C., et al. (2020) Remdesivir for the treatment of Covid-19 — Preliminary report. New England Journal of Medicine 383: 1813-1826 https://doi.org/10.1056/NEJMoa2007764 | Google Scholar
- Borba M. G. S., Val F. F. A., Sampaio V. S.. (2020) Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: A randomized clinical trial. JAMA Network Open 3: e208857 https://doi.org/10.1001/jamanetworkopen.2020.8857 | Google Scholar
- Caly L., Druce J. D., Catton M. G., Jans D. A., Wagstaff K. M.. (2020) The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 . Antiviral Research 178: 104787 https://doi.org/10.1016/j.antiviral.2020.104787 | Google Scholar
- Cao B., Wang Y., Wen D.. (2020) A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. New England Journal of Medicine 382: 1787-1799 https://doi.org/10.1056/NEJMoa2001282 | Google Scholar
- Cavalli G., De Luca G., Campochiaro C.. (2020) Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: A retrospective cohort study. The Lancet Rheumatology 2: e325-e331 https://doi.org/10.1016/S2665-9913(20)30127-2 | Google Scholar
- Chorin E., Dai M., Shulman E.. (2020) The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nature Medicine 26: 808-809 https://doi.org/10.1038/s41591-020-0888-2 | Google Scholar
- CredibleMeds.. 2020 Combined list of drugs that prolong QT and/or cause torsades de pointes (TDP).
- El-Senousey H., Chen B., Wang J., Atta A., Mohamed F., Nie Q., et al. (2018) Effects of dietary vitamin C, vitamin E, and alpha-lipoic acid supplementation on the antioxidant defense system and immune-related gene expression in broilers exposed to oxidative stress by dexamethasone. Poultry Science 97: 30-38 https://doi.org/10.3382/ps/pex281 | Google Scholar
- Eltahan NH, Elsawy NH, Abdelaaty KM. (2024) Atorvastatin for reduction of 28-day mortality in severe and critical COVID-19 patients: a randomized controlled trial. Respir Res. 25: 97 https://doi.org/10.1186/s12931-024-02732-2 | Google Scholar
- Fang L., Karakiulakis G., Roth M.. (2020) Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection?. The Lancet Respiratory Medicine 8: e21 https://doi.org/10.1016/S2213-2600(20)30116-8 | Google Scholar
- Fedson D. S., Opal S. M., Rordam O. M.. (2020) Hiding in plain sight: An approach to treating patients with severe COVID-19 infection. mBio 11: e00398-20 https://doi.org/10.1128/mBio.00398-20 | Google Scholar
- Food and Drug Administration.. 2020 FDA advises patients on use of non-steroidal anti-inflammatory drugs (NSAIDs) for COVID-19.
- Gautret P., Lagier J.-C., Parola P., Meddeb L., Mailhe M., Doudier B., et al. (2020) Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial. International Journal of Antimicrobial Agents 56: 105949 https://doi.org/10.1016/j.ijantimicag.2020.105949 | Google Scholar
- Goldman J. D., Lye D. C., Hui D. S.. (2020) Remdesivir for 5 or 10 days in patients with severe COVID-19. New England Journal of Medicine 383: 1827-1837 https://doi.org/10.1056/NEJMoa2015301 | Google Scholar
- Hassan AH, Hegazy SK, Radwan ST.. (2022) Clinical study to evaluate the possible efficacy and safety of antibodies combination (casirivimab and imdevimab) versus standard antiviral therapy as antiviral agent against Corona virus 2 infection in hospitalized COVID-19 patients. medRxiv https://doi.org/10.1101/2022.08.20.22279020 | Google Scholar
- Hassan AH, Hegazy SK, Radwan ST.. (2023) Clinical study to evaluate the possible efficacy and safety of antibodies combination (casirivimab and imdevimab) versus standard antiviral therapy as antiviral agent against Corona virus 2 infection in hospitalized COVID-19 patients. Res Square. https://doi.org/10.21203/rs.3.rs-1991618/v2 | Google Scholar
- Hegazy SK, Hassan AH.. (2024) Comparing the efficacy of remdesivir, favipiravir, and casirivimab and imdevimab on duration of hospitalization and ICU stay of hospitalized COVID-19 patients. Life Sci Res Commun. 1: 21-30 https://doi.org/10.21203/rs.3.rs-1991618/v2 | Google Scholar
- Hegazy SK, Tharwat S, Hassan AH.. (2023) Clinical study to compare the efcacy and safety of casirivimab and imdevimab, remdesivir, and Favipiravir in hospitalized COVID-19 patients. J Clin Virol Plus. 3: 100151 https://doi.org/10.1016/j.jcvp.2023.100151 | Google Scholar
- Hegazy SK, Tharwat S, Hassan AH.. (2023) Comparing the efficacy of regen-cov, remdesivir, and favipiravir in reducing invasive mechanical ventilation need in hospitalized COVID-19 patients. World J Clin Cases. 11: 6105 https://doi.org/10.1016/j.jcvp.2023.100151 | Google Scholar
- Hegazy SK., Tharwat S, Hassan AH.. (2023) Study to compare the effect of casirivimab and imdevimab, remdesivir, and favipiravir on progression and multi-organ function of hospitalized COVID-19 patients. Open Medicine vol. 18: 20230768 https://doi.org/10.1515/med-2023-0768 | Google Scholar
- Horby P., Lim W. S., Emberson J.. (2020) Effect of dexamethasone in hospitalized patients with COVID-19: Preliminary report. medRxiv https://doi.org/10.1101/2020.06.22.20137273 | Google Scholar
- Kaiser U. B., Mirmira R. G., Stewart P. M.. (2020) Our response to COVID-19 as endocrinologists and diabetologists. Journal of Clinical Endocrinology and Metabolism 105: dgaa148 https://doi.org/10.1210/clinem/dgaa148 | Google Scholar
- Kim U. J., Won E.-J., Kee S.-J., Jung S.-I., Jang H.-C.. (2016) Combination therapy with lopinavir/ritonavir, ribavirin and interferon-α for Middle East respiratory syndrome. Antiviral Therapy 21: 455-459 https://doi.org/10.3851/IMP3002 | Google Scholar
- Kuba K., Imai Y., Rao S., Gao H., Guo F., Guan B., et al. (2005) A crucial role of angiotensin-converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nature Medicine 11: 875-879 https://doi.org/10.1038/nm1267 | Google Scholar
- Molina J. M., Delaugerre C., Le Goff J., Mela-Lima B., Ponscarme D., Goldwirt L., et al. (2020) No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Médecine et Maladies Infectieuses 50: 384 https://doi.org/10.1016/j.medmal.2020.03.006 | Google Scholar
- Patel A. B., Verma A.. (2020) COVID-19 and angiotensin-converting enzyme inhibitors and angiotensin receptor blockers: What is the evidence?. JAMA 323: 1769-1770 https://doi.org/10.1001/jama.2020.4812 | Google Scholar
- Press Release.. PR Newswire.. 2020 Tocilizumab improves significantly clinical outcomes of patients with moderate or severe COVID-19 pneumonia.
- Richardson P., Griffin I., Tucker C.. (2020) Baricitinib as potential treatment for 2019-nCoV acute respiratory disease. The Lancet Rheumatology 2: e30-e31 https://doi.org/10.1016/S2665-9913(20)30016-2 | Google Scholar
- Rosa S. G. V., Santos W. C.. (2020) Clinical trials on drug repositioning for COVID-19 treatment. Revista Panamericana de Salud Pública 44: e40 https://doi.org/10.26633/RPSP.2020.40 | Google Scholar
- Sachse G., Willms B.. (1980) Efficacy of thioctic acid in the therapy of peripheral diabetic neuropathy. Hormone and Metabolic Research Supplement Series 9: 105-107 https://doi.org/10.26633/RPSP.2020.40 | Google Scholar
- Hegazy Sahar K.. (2024) The efect of combination treatment with casirivimab and imdevimab versus standard antiviral therapy on clinical outcomes in hospitalized COVID-19 patients. Discover 1 https://doi.org/10.1007/s44337-024-00045-3 | Google Scholar
- Savarino A., Boelaert J. R., Cassone A., Majori G., Cauda R.. (2003) Effects of chloroquine on viral infections: An old drug against today’s diseases. The Lancet Infectious Diseases 3: 722-727 https://doi.org/10.1016/S1473-3099(03)00806-5 | Google Scholar
- Sciascia S., Aprà F., Baffa A.. (2020) Pilot prospective open, single-arm multicentre study on off-label use of tocilizumab in patients with severe COVID-19. Clinical and Experimental Rheumatology 38: 529-532 https://doi.org/10.1016/S1473-3099(03)00806-5 | Google Scholar
- Shakoory B., Carcillo J. A., Chatham W. W.. (2016) Interleukin-1 receptor blockade is associated with reduced mortality in sepsis patients with features of macrophage activation syndrome: Reanalysis of a prior Phase III trial. Critical Care Medicine 44: 275-281 https://doi.org/10.1097/CCM.0000000000001402 | Google Scholar
- Sheahan T. P., Sims A. C., Leist S. R., Schäfer A., Won J., Brown A. J., et al. (2020) Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV. Nature Communications 11: 222 https://doi.org/10.1038/s41467-019-13940-6 | Google Scholar
- Tsang K., Zhong N. S.. (2003) SARS: Pharmacotherapy. Respirology 8: S25-S30 https://doi.org/10.1046/j.1440-1843.2003.00522.x | Google Scholar
- Tibullo D., Volti G. L., Giallongo C., Grasso S., Tomassoni D., Anfuso C. D., et al. (2017) Biochemical and clinical relevance of alpha lipoic acid: Antioxidant and anti-inflammatory activity, molecular pathways and therapeutic potential. Inflammation Research 66: 947-959 https://doi.org/10.1007/s00011-017-1079-8 | Google Scholar
- Vincent M. J., Bergeron E., Benjannet S., Erickson B. R., Rollin P. E., Ksiazek T. G., et al. (2005) Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virology Journal 2: 69 https://doi.org/10.1186/1743-422X-2-69 | Google Scholar
- Wang X., Guo X., Xin Q.. (2020) Neutralizing antibody responses to SARS-CoV-2 in COVID-19 inpatients and convalescent patients. medRxiv. https://doi.org/10.1101/2020.04.22.20075091 | Google Scholar
- World Health Organization.. 2020 Situation report – 193.
- Wu C. J., Jan J. T., Chen C. M., Hsieh H. P., Hwang D. R., Liu H. W., et al. (2004) Inhibition of severe acute respiratory syndrome coronavirus replication by niclosamide. Antimicrobial Agents and Chemotherapy 48: 2693-2696 https://doi.org/10.1128/AAC.48.7.2693-2696.2004 | Google Scholar
- Wu Y.-H., Tseng C.-P., Cheng M.-L., Ho H.-Y., Shih S.-R., Chiu D. T.-Y., et al. (2008) Glucose-6-phosphate dehydrogenase deficiency enhances human coronavirus 229E infection. Journal of Infectious Diseases 197: 812-816 https://doi.org/10.1086/528377 | Google Scholar
- Yamamoto N., Yang R., Yoshinaka Y., Amari S., Nakano T., Cinatl J., et al. (2004) HIV protease inhibitor nelfinavir inhibits replication of SARS-associated coronavirus. Biochemical and Biophysical Research Communications 318: 719-725 https://doi.org/10.1016/j.bbrc.2004.04.083 | Google Scholar
- Ye M., Fu D., Ren Y., Wang F., Wang D., Zhang F., et al. (2020) Treatment with convalescent plasma for COVID-19 patients in Wuhan, China. Journal of Medical Virology 92: 1858-1865 https://doi.org/10.1002/jmv.25882 | Google Scholar
- Zeng Q., Yu Z., Gou J.. (2020) Effect of convalescent plasma therapy on viral shedding and survival in COVID-19 patients. Journal of Infectious Diseases 222: 38-43 https://doi.org/10.1093/infdis/jiaa228 | Google Scholar
- Zhang L., Liu Y.. (2020) Potential interventions for novel coronavirus in China: A systematic review. Journal of Medical Virology 92: 479-490 https://doi.org/10.1002/jmv.25707 | Google Scholar
- Zhang X. W., Yap Y. L.. (2004) Old drugs as lead compounds for a new disease? Binding analysis of SARS coronavirus main proteinase with HIV, psychotic and parasite drugs. Bioorganic and Medicinal Chemistry 12: 2517-2521 https://doi.org/10.1016/j.bmc.2004.03.035 | Google Scholar



